TL;DR Patients cross many systems (HIS, LIS, PACS, billing, TPA). DPDP expects one coherent notice-and-consent story. Bolting privacy onto a single vendor’s EMR leaves labs, imaging, and payers structurally “out of scope.” Kavach is modeled as a cross-cutting accountability layer—APIs, webhooks, optional in-browser assist—not a replacement EMR module.
Patients do not experience “one system.” They move from registration to OPD, into labs and imaging, through pharmacy, sometimes wards and theatres, and often insurance desks. The Digital Personal Data Protection Act expects notices, lawful grounds, and accountability to stay coherent across that journey—not fragmented across vendor upgrades and module silos.
This article expands on our healthcare solution overview for CMIOs, IT heads, and compliance mapping DPDP to real hospital operations. Below: Mermaid topology + flow diagrams, a vector sketch of the integration pattern, and the full department × data × notice matrix.
What you’ll take away
- Why treating consent as “just another HIS module” creates blind spots under DPDP.
- A department-by-department view of where personal data appears and which notice themes usually matter.
- How elective and emergency paths differ for documentation (governance—not a clinical protocol).
- How a cross-cutting layer integrates LIS, RIS/PACS, billing, and portals without folding privacy into a single supplier’s roadmap.
Reference topology (vector sketch)
ASCII-friendly boxes · not a network diagram to scale
Consent management cannot be only an HIS module
Your Hospital Information System (HIS / EMR) is essential—but it is only one system. Laboratory (LIS), imaging (RIS/PACS), pharmacy, OT modules, insurance/TPA portals, and your public website each hold or move patient data. If consent is trapped inside the HIS vendor’s roadmap, you risk inconsistent notice versions, departments that never saw the same policy pack, and patients who cannot see one truthful timeline.
Kavach is deliberately separate: notices, purposes, consent, withdrawal, and rights requests are versioned and auditable, then shared back to every system that needs to know—without making any single clinical supplier the owner of India’s data-protection evidence.
Integrate via APIs and webhooks so orders, identifiers, and consent state stay aligned. Where the HIS cannot change quickly, browser-based assistance can surface notices in-context on the screens staff already use—without migrating the whole EMR.
When the HIS vendor owns consent UX and storage, every LIS/PACS migration and every TPA API change becomes a negotiation. Kavach inverts that: your hospital owns the policy pack and evidence; clinical systems consume it.
Where personal data shows up—department by department
Use this map to align clinical reality with DPDP expectations. At each stop, teams should know what is collected, for which purpose, and what the patient can later see or challenge. Wording is operational, not legal advice; your counsel confirms lawful grounds in edge cases.
| Department / area | Typical personal data activity | Notice and consent themes that usually matter |
|---|---|---|
| Front desk / registration | Demographics, identifiers, insurance cards, contact preferences, visit creation. | Facility privacy notice; identity verification; communications (SMS, WhatsApp, email); optional marketing separation. |
| OPD | Clinical history, examination notes, orders, referrals. | Care delivery; sharing with diagnostics and consultants; teleconsult if used; access by treating team only. |
| Laboratory | Specimen ID, test panels, results, reflex testing, QC data linked to patients. | Test-specific processing; referral lab sharing; retention for clinical and statutory periods; result delivery channels. |
| Radiology / imaging | Imaging orders, images, reports, dose records, sharing for second opinion. | Imaging purposes; teleradiology; external archive; patient copy of report. |
| Pharmacy | Prescriptions, dispensing, counselling notes, home delivery addresses. | Medication fulfilment; adherence programmes if any; delivery partners. |
| IPD / wards | Admission notes, nursing charts, vitals, family contact, discharge summary. | Inpatient care; visitors; transfers between units; discharge instructions. |
| Operation theatre | Surgical consent artefacts, anaesthesia records, implant traceability. | Procedure-specific informed consent; blood/blood product rules; photography if applicable. |
| Emergency / trauma | Triage, stabilisation, next-of-kin, police / medico-legal context, rapid orders. | Often legitimate uses and urgency; deferred detailed consent when clinically appropriate; documentation for later reconciliation. |
| ICU | Continuous monitoring, critical care notes, family updates, organ-support decisions. | Intensive care delivery; family communication; research biospecimens only with proper governance. |
| Daycare / dialysis | Short-stay treatment records, vascular access, recurring schedules. | Recurring treatment purposes; long-running retention; partner dialysis chains if referred. |
| Physiotherapy | Functional assessment, therapy plans, progress notes, images of movement. | Rehabilitation; outcome tracking; referral back to ortho/neuro teams. |
| Dental | Oral imaging, treatment plans, billing for procedures. | Dental care-specific notices; cosmetic vs therapeutic distinction where relevant. |
| Eye / ophthalmology | Visual tests, surgical planning, device implant registers. | Ophthalmic care; device traceability; research registries if opted-in. |
| Insurance / TPA desk | Claims packets, pre-auth, settlement correspondence. | Claims processing; fraud prevention limits; data minimisation to payer; separate from marketing use. |
Many vendors, one consent story
Hospitals rarely run a closed loop: samples go to external labs, images may be read off-site, claims cross to TPAs. The pattern that scales: integrate via APIs and webhooks so orders, identifiers, and consent state stay aligned. When an order “leaves the building,” bind notice and purpose before data crosses—then log outcomes so the patient portal reflects the same narrative as internal evidence.
Whether the partner is a reference lab, a teleradiology hub, or a payer portal, the pattern is the same: do not forward everything—forward what is lawful for that purpose, log it, and keep the DP-facing view consistent.
Elective journey and recurring purposes
Most non-emergency visits follow a recognisable path. The diagram below is a happy-path schematic—your triage rules may branch earlier.
At registration, baseline facility notices and communication preferences apply. At order time, diagnostics-specific purposes attach to lab/imaging worklists. At pharmacy, fulfilment and delivery partners are covered. If the patient is admitted, inpatient and surgical consent artefacts layer on without discarding upstream captures.
Purposes that recur: clinical care; diagnostics; billing and claims (minimum necessary to TPAs); quality and safety; optional uses (marketing, fundraising, research) kept separate and clearly opted-in.
For medical leadership: Kavach does not replace clinical consent forms for procedures. It ensures the data-protection story—what personal data left each step, under which notice, and how the patient can access or withdraw non-essential uses—stays consistent with DPDP expectations.
Emergency and trauma: care first, defensible documentation after
Emergency departments handle patients who may be unconscious or in distress. Lawful grounds are not identical to a calm registration desk: certain processing may proceed on bases other than fresh explicit consent—for example medical treatment in an emergency—subject to your legal interpretation and protocols.
Kavach supports documentation and later reconciliation: what was processed under urgency, when the patient was informed, and how optional uses were kept separate. It does not ask clinicians to delay stabilisation for a privacy form; it asks the hospital to be defensible after the fact.
What hospital counsel and CMIO teams usually want: a clear line between essential emergency care and optional processing; protocols for substitute decision-makers and children; audit fields showing when detailed privacy information was provided once the patient can receive it; consistency between HIS/LIS behaviour and what the patient portal shows.
This article is not legal advice. Emergency processing depends on facts, sector rules, and how the DPDP Act applies to your institution.
Patient-facing transparency
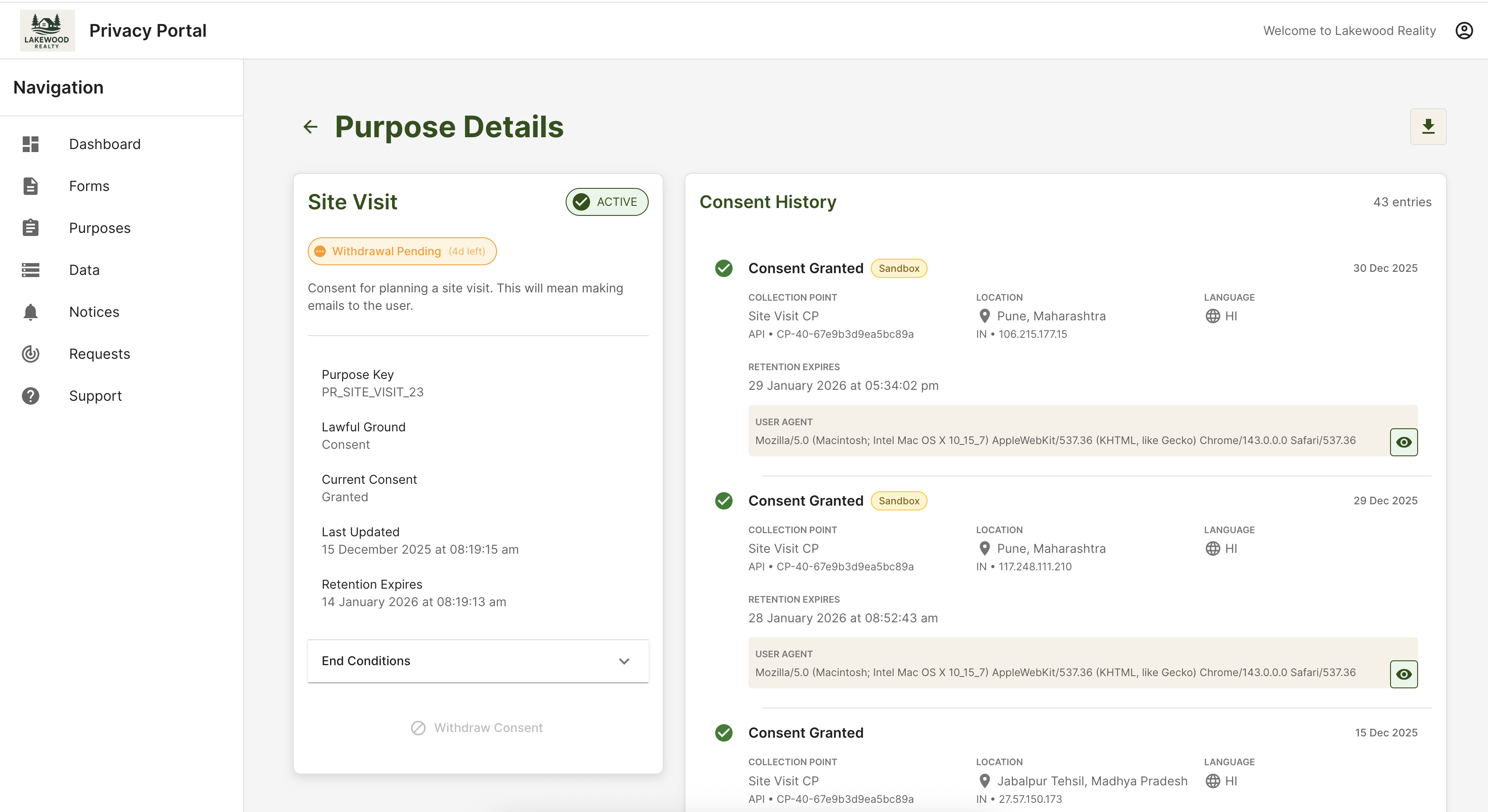
Your Data Principal Portal should mirror what compliance exports internally: active purposes, consent history, withdrawals, and open requests—in language patients and families understand, so “the hospital told me something different” disputes shrink.
Below is a representative Kavach portal view (purpose detail + consent history timeline), hosted on this site as /images/DP_portal.png. Demo tenants may show placeholder organisation names in the chrome; your production skin would use your hospital’s branding.
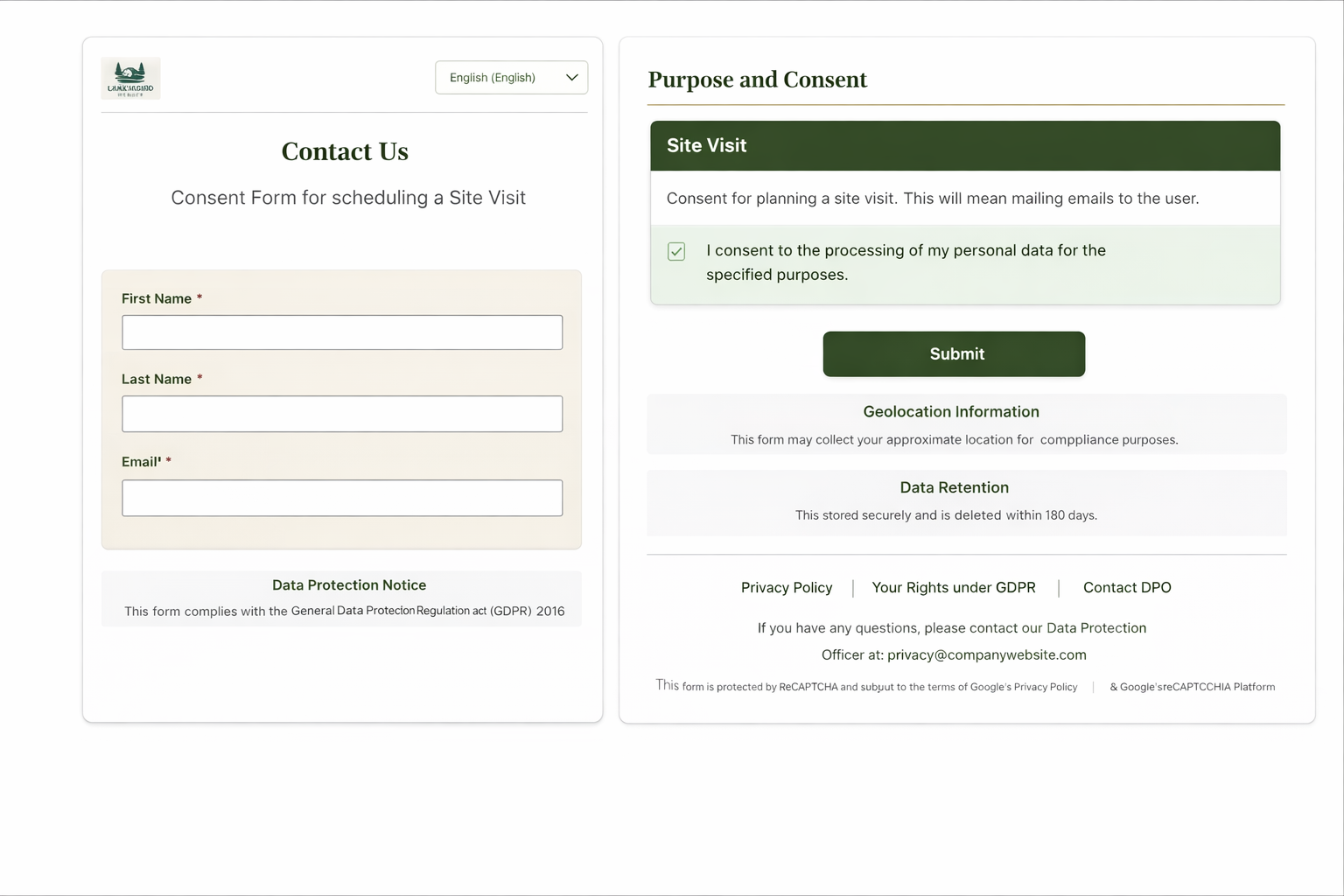
Staff-facing capture can use the same notice and purpose pattern at registration or kiosks:
For a fuller walkthrough, see the healthcare solution page or book a briefing.
Related links
Kavach provides software for notices, consent, patient rights workflows, and audit evidence. Implementation and lawful basis for specific processing must be confirmed with your legal and clinical governance teams.